Revision Shoulder Surgery
Basics of failed shoulder surgery and revision shoulder surgery
A shoulder surgery has failed when it does not achieve the expectations of the patient and the surgeon. Failure can result from stiffness, weakness, instability, pain or failure to heal, and from complications such as infection or nerve injury. Every surgery has a risk of failure whether it is an operation for dislocation, rotator cuff tear, arthritis or fracture. Because fracture fixation, Bankart repairs, rotator cuff repairs, shoulder joint replacements and reverse total shoulders are performed commonly, a substantial number of patients have experienced these failures.
When a patient experiences a failure of shoulder surgery, patients should consult a surgeon experienced in the evaluation and management of these conditions. Even though the problem may seem to be pain it is important to identify the likely mechanical causes of the failure in that a mechanical cause may be repairable. If a shoulder surgery has resulted in stiffness or limited range of motion, stretching exercises or a second surgery to release adhesions may be helpful. If a rotator cuff repair has resulted in weakness or catching and popping on shoulder movement, a ‘smooth and move,’ or possibly, a re-repair may be considered. If a shoulder arthroscopy has resulted in stiffness, chondrolysis may have resulted. If a Bankart repair or surgery for shoulder dislocations has resulted in persistent instability or limited motion, a revision repair or removal of adhesions may be of benefit. If a conventional shoulder replacement for shoulder arthritis or a reverse total shoulder replacement has become stiff, unstable, or if the artificial components have become loose, a revision surgery to rebalance the tissues and to re-secure the humeral and glenoid replacement parts may restore comfort and function. Finally, if surgery for a fracture has resulted in stiffness or non-healing of the bone, removal of scar tissue and re-fixation of the fracture may improve the result.
The evaluation begins with a history of the shoulder problem starting before the surgery, a review of the report from the surgery, a physical examination, high quality X-rays, and a complete assessment of the overall health of the patient. This evaluation enables the experienced revision shoulder surgeon to discuss the different treatment options with the patient, including the potential risks and benefits.
Revision shoulder surgery is rarely an emergency. Substantial thought should go into considering the pros and cons of additional surgery and when and where it can be best performed.
Revision surgery for failed total shoulder replacement arthroplasty
When a shoulder replacement fails to result in satisfactory restoration of comfort and function the surgeon and the patient are challenged to determine the most likely causes and how the failure can be best managed. It is always important to keep in mind the wide range of potential causes of joint replacement failure. These have been well characterized by our shoulder fellows Hasan and Franta.
While pain is often a presenting complaint, we try to determine as well if the shoulder is stiff, unstable, weak, or crepitant. Each of these mechanical characteristics provides clues to what may be going on with the joint. For example, a shoulder arthroplasty may be stiff because of adhesions (scar), blocking osteophytes (bone spurs), or overstuffing (too large prosthetic components). Weakness may result from rotator cuff failure, subscapularis detachment, nerve injury, deltoid detachment, or disuse atrophy of the muscles. Instability may result from suboptimal positioning of the components, component loosening, or soft tissue imbalance. Crepitation, clicking, or clunking may result from component loosening, soft tissue ingrowth, loose bodies within the joint, or joint surface irregularities.
As pointed out by Hasan and Franta, infection is an ever present concern in painful shoulder arthroplasties. Trying to better understand the causes, prevention, diagnosis, and treatment of shoulder joint replacement infections has become a major interest of mine. Please consult Dr. Frederick Matsen’s shoulder arthritis blog for some of our late-breaking discoveries. For now, suffice it to say that shoulder infections most often present themselves only by shoulder pain – the ‘traditional’ evidences of infection, such as fever, chills, redness, swelling, elevated white blood cell count, elevated sedimentation rate, and elevated C reactive protein, are usually absent.
A thorough evaluation of the patient and the shoulder are essential before considering the best treatment. A careful history is needed to determine the original diagnosis, the initial result of the joint replacement, the onset of discomfort or loss of function, any injuries, dental or other procedures that may have introduced infection, and any intercurrent diseases. The physical exam must seek evidence of stiffness, weakness, instability, or crepitance. Finally, high quality x-rays are needed to look for the position and relationship of the prosthetic components as well as evidence of loosening or wear of these components.
Principles of Revision Surgery
Considering Revision Surgery
Revision shoulder surgery calls on judgment, experience, and technical skills that are an order of magnitude greater than for primary shoulder surgery. There are ten questions that we ask before taking on a revision case:
- Do we have sufficient past records on this case?
- Is the problem a mechanical one that is clearly identified (as opposed to a problem that is manifest as pain or frustration without a clear mechanical cause)?
- Is there a nonoperative approach to the problem?
- Is the mechanical problem treatable?
- Is the patient of sufficient mental and physical health and strength to undergo a revision surgery – are the patient’s metabolism and the skin over the shoulder ready for another surgery? has the patient ceased smoking? are alcohol consumption and pain medication use under control?
- Are the patient’s expectations reasonable?
- Does the patient fully understand the risks and possible outcomes of surgery, including the anticipated incision and the possibility that infection may be encountered?
- Do we need a consultation to help define the cause and treatment of the problem?
- Are we the best surgeons to carry out the revision surgery?
- Do we have the right tools and team to carry out the surgical revision?
To answer these questions we seek the following information prior to considering a surgical revision:
- An understanding of the patient’s status prior to the index procedure.
- Previous operative notes, including information on the type, manufacturer, and size of implants.
- An assessment of the legal and insurance aspects of the case.
- Knowledge of the medical status of the patient including:
- Health conditions that may affect the patient’s surgery
- Current medications, including pain medications and dosages
- The amount of nicotine and alcohol currently being consumed
- The psychological status of the patient
- The vocational status of the patient
- The social situation and support systems for the patient
- Current laboratory values, including a CBC sedimentation rate and serum albumin.
- High quality anteroposterior and axillary radiographs as well as an anteroposterior radiograph of the entire humerus.
- Completed Simple Shoulder Test and Short Form-36 Questionnaires.
- A physical examination of the neck and shoulder, including the location of skin incisions and the health of the skin in the areas of possible incision.
- An EMG if there is concern about radiculopathy or neuropathy.
- Consent for bone autograft tendon autograft or allograft as necessary.
- Medicine, anesthesiology, and pain service consults as necessary.
- Urine analysis to screen for drug alcohol and nicotine if indicated.
Common Causes of shoulder arthroplasty failure
Before embarking on a surgical revision of a shoulder arthroplasty it is important to determine the nature of the patient’s problems. The following is a list of the common causes of shoulder arthroplasty failure:
- Infection
- Reaction to polyethylene or polymethylmethacrylate
- Fracture
- Stiffness
- Poor rehabilitation
- Unwanted bone
- Tuberosity malunion
- Overstuffing of the joint
- Instability
- Anterior
- Subscapularis deficiency
- Glenoid component anteversion
- Tuberosity nonunion
- Supraspinatus/infraspinatus defect
- Humeral component anteversion or anterior head offset
- Insufficient anterior glenoid bone
- Posterior
- Glenoid component retroversion
- Posterior cuff defect
- Excessive humeral component retroversion or posterior head offset
- Insufficient posterior glenoid bone
- Superior
- Loss of rotator cuff
- Loss of coracoacromial arch
- Anterior
- Weakness
- Reduced muscle strength
- Subscapularis deficiency
- Supraspinatus/infraspinatus deficiency
- Tuberosity nonunion or malunion
- Deltoid detachment
- Nerve injury
- Humeral component
- Malpositioned
- Loose
- Glenoid
- Bone eroded (hemiarthroplasty)
- Component malpositioned
- Component loose
Preparing for Revision Surgery
The history and previous records are reviewed to learn the status of the patient and shoulder prior to the index arthroplasty. What were the details of the reconstruction, including the manufacturer, model, and size of the prostheses? How was the rehabilitation conducted? Is there evidence of infection or allergic reaction? Has there been an intercurrent injury? What is the patient’s status with respect to nutrition, pain, medications, smoking, alcohol, and other concurrent health conditions?
The workup includes a detailed examination of the motion, stability, strength, and smoothness of the shoulder.
EMG’s and nerve conduction studies, CT scans, and expert sonography may be useful in evaluating the nerve function bone and rotator cuff respectively.
Laboratory studies include a CBC sedimentation rate and serum albumin.
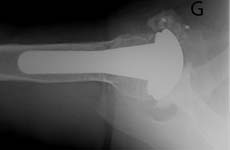
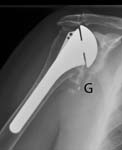
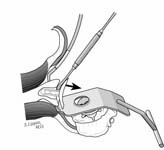
Radiographs include an anteroposterior view in the plane of the scapula an axillary view and a full humeral view all of high quality. Here is an AP view and an axillary view showing a glenoid component that has completely loosened from the bone and is floating free within the joint (two white dots near the letter “G”). [See Figures 19 20]
In cases of instability, examination under fluoroscopy may be useful. The radiographic evaluation must confirm the type and size of components, their position, and the nature of their fixation to bone. The preoperative plan must include a definitive plan for removal of the glenoid and humeral components, should this prove necessary, as well as a plan for reconstruction of the humerus and the glenoid after prosthesis removal. If removal of a cemented humeral component may be necessary, it is essential to have a full set of cement removal tools, a high speed saw capable of cutting a prosthetic stem fluoroscopy, and long stem prostheses of all possible sizes. The possible need for bone and tendon graft is also anticipated.
Click an image below to enlarge and view in the gallery.
Surgical Considerations and General Principles of Revision Surgery
Before the anesthetic, the patient’s consent is checked to make sure it is complete. The instrument and implant inventory are verified for possible variations on the preoperative plan, including the need to modify a prosthesis or to possibly use a special implant (e.g. a long stemmed humeral implant in case of shaft fracture).
The patient is anesthetized, positioned, prepped, and draped in a manner that anticipates all possible variations on the surgical plan. The entire forequarter is prepped so that incisions can be made anteriorly, posteriorly, or distally as needed. The arm is draped so that it can be moved freely. Ipsilateral, iliac, crest, and hamstring autograft donor sites are prepped if their possible need is anticipated.
Preoperative antibiotics are not administered. Prophylactic antibiotics are administered only after specimens are collected for culture and sensitivity testing.
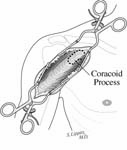
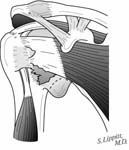
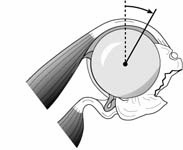
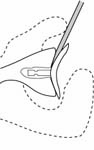
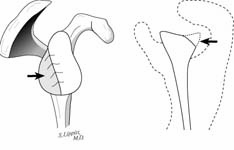
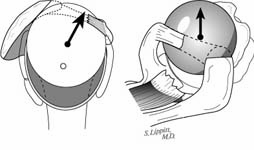
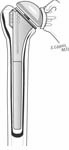
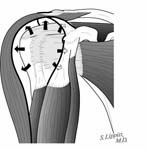
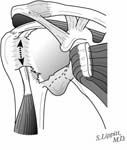
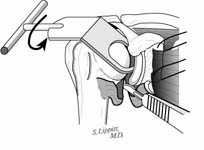
The incision is made in a manner that provides optimal access to the mechanical problem and, if possible, incorporates or respects previous skin incisions. The possible need for extending the incision is anticipated. As shown below, the coracoid is an important landmark when the normal anatomy of the deltopectoral interval is scarred from previous surgery through the anterior approach.
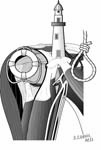
The surgical approach is conducted carefully to protect and preserve the deltoid, the rotator cuff, and the neurovascular structures about the shoulder – each of which may have been altered by previous surgery. The coracoid process serves as a lighthouse for proper orientation in a scarred shoulder. It divides the lateral (safe side) from the medical side (suicide) where the brachial plexus and vascular structures are located (See Figures 1 and 2).
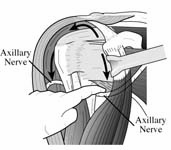
The humeroscapular motion interface is entered and all adhesions lysed. The axillary nerve is identified and protected.
Specimens are collected for aerobic, anaerobic, and fungal culture sensitivity and gram stains. Frozen and permanent sections are obtained of any tissue suspicious for inflammation, infection, or neoplasm. Prophylactic antibiotics are given intravenously at this point.
Click an image below to enlarge and view in the gallery.
Types of Revision Surgery for Shoulder Replacement Arthroplasty
Revision Surgery due to Stiffness – Soft Tissue Release
Before embarking on a surgical revision to regain motion, it is important to determine the cause of the residual stiffness. Previous operative notes are reviewed to discover if the joint was intentionally tightened and, if so, how this was carried out. Particular note is made of whether the subscapularis was advanced lateral to the bicipital groove and whether thermal or laser capsular cauterization was carried out. Excellent anteroposterior and axillary radiographs are needed to exclude bony or articular causes of shoulder stiffness. The location of hardware and other implants is determined.
Prior to prepping and draping the shoulder, the ranges of flexion, cross body adduction, internal and external rotation in 90 degrees of abduction, and external rotation at the side, as well as the excursion on posterior drawer testing are recorded for both shoulders. [See Figure 3]
The surgical procedure is performed sequentially with the range of motion being reexamined after each step of the release. The procedure may be concluded when the desired range is achieved.
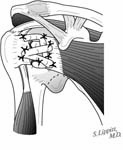
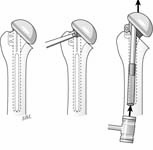
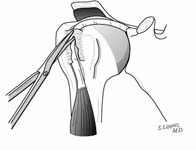
The initial step is to completely free the humeroscapular motion interface.
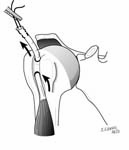
Any prominent suture, suture anchors, hardware, bone, or soft tissue is resected from the proximal humerus to assure smooth passage within the coracoacromial arch. A nerve-to-nerve release is performed. [See Figure 4]
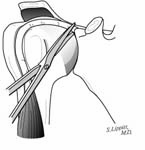
The second step is to incise the coracohumeral ligament from around the coracoid process. [See Figure 5]
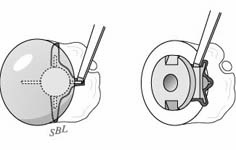
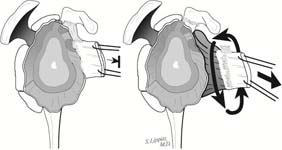
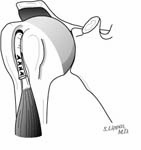
Next, the subscapularis is identified and incised from its humeral attachment, preserving maximal length. A 360-degree release of the subscapularis and anterior capsule is carried out assuring that the subscapularis moves freely with respect to the coracoid, the glenoid lip, the inferior capsule, and the axillary nerve. [See Figure 6]
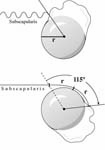
In this release the anterior capsule is incised just lateral to the labrum leaving the labrum on the bony glenoid to maintain the glenoid concavity. After the release the subscapularis should have a nice ‘bounce’ when traction is applied to it. Recall that if the subscapularis is to allow a range of rotation of 115 degrees (two radians), it must have an excursion of twice the radius of the humeral head. Thus, if a humeral head has a radius of 25 mm, a subscapularis excursion of 50 mm will allow a 115-degree range of internal and external rotation. [See Figure 7]
After this complete release additional subscapularis lengthening is rarely needed. However, if additional lengthening of the subscapularis tendon is needed, an inside-out coronal plane Z-plasty is considered if there is adequate thickness of the capsule and tendon. Sometimes a hamstring autograft is used to robustly connect the subscapularis to the lesser tuberosity with additional tendon length. It must be noted, however, that adding tendon length does not increase the functional excursion of the muscle. [See Figure 8]
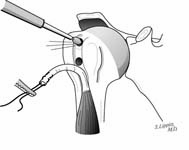
If the long head tendon of the biceps does not slide freely in the bicipital groove, the adhesions in the groove are released. If freedom cannot be achieved, the tendon is incised at its insertion to the supraglenoid tubercle and tenodesed to the proximal humerus in its groove. [See Figures 9-14]
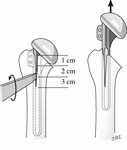
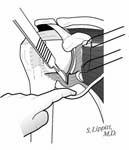
The next step is protecting the axillary nerve with the non-dominant index finger to release the inferior capsule from the inferior glenoid labrum. [See Figure 15]
Inserting a humeral head retractor into the joint and rotating its inferior aspect away from the glenoid puts the inferior capsule under tension facilitating this release. The anterior and posterior bands of the inferior glenohumeral ligament are released. Exposure of the origin of the long head of the triceps signals a complete release. [See Figure 16]
The articular aspect of the joint is inspected and any bony prominences that potentially block motion are resected. Note is made of the condition of the glenoid and humeral joint surfaces.
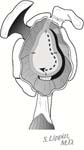
Finally, the posterior capsule is released from the posterior glenoid labrum. The capsule is placed under tension by rotating the humeral head retractor away from the glenoid first inferiorly and then superiorly. [See Figure 17-18]
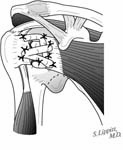
At the conclusion of the procedure, the shoulder is put through a full range of motion. The subscapularis tendon is closed robustly to the lesser tuberosity so that immediate postoperative motion maintaining exercises can be implemented.
Click an image below to enlarge and view in the gallery.

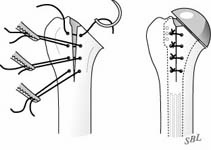
Revision surgery due to weakness
If weakness is an issue after shoulder arthroplasty, it may be due to:
- insufficient time and effort at rehabilitation (recognizing that shoulder joint replacement is performed on shoulders that have been ‘out of action’ for a long period of time with some disuse atrophy of the muscles),
- technical problems with the arthroplasty,
- nerve injuries,
- rotator cuff defects, or
- subscapularis tendon detachment.
In a technically well-done joint replacement and in the absence of some intercurrent injury, persistence of the rehabilitation program will usually lead to progressively more strength and function. If this fails the shoulder should be scrutinized for evidence of other causes of weakness as listed above.
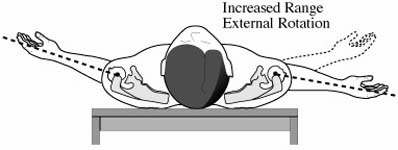
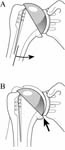
Failure of the subscapularis reattachment may result from excessive early loading of the repair or from poor quality of the tendon. [See Figure 21]
This situation is suggested by weakness of internal rotation and an increased range of external rotation. [See Figure 22]
If there has been an acute injury causing the disruption, a re-repair may be indicated. Otherwise, a reconstruction with a tendon graft may be helpful in restoring some of the function of the subscapularis. [See Figure 23]
Click an image below to enlarge and view in the gallery.
Revision surgery due to infection
Surgical cultures obtained from shoulders requiring revision of a prior arthroplasty due to pain, stiffness, or loosening are frequently positive despite the absence of preoperative evidence of joint sepsis. Often these cultures are positive for organisms such as Propionibacterium acnes and Coagulase-negative Staphylococci that are less commonly recovered from revision surgery in other sites of joint replacement such as the hip or knee.
These organisms are distinguished by:
- their presence on normal skin,
- their failure to engender systemic manifestations of infection such as elevated C reactive protein sedimentation rate and white blood cell count,
- their failure to produce local clinical evidence of infection such as redness swelling and tenderness,
- the low yield of cultures of joint aspiration, and
- the difficulty in eradicating infections with these organisms.
We recently completed a study regarding cultures obtained at the time of revision arthroplasty. The abstract from this as yet unpublished study is reproduced below.
Background: P. acnes is a gram-positive anaerobic bacterium not infrequently cultured at the time of revision shoulder arthroplasty even when common clinical and laboratory signs of sepsis are absent. Because several weeks of culture incubation can be required to recover this organism, clinical decisions regarding the type of revision surgery and the post-operative antibiotic treatment program must be made before the culture results are finalized. Our goal is to seek prognostic information that would help guide necessary decision-making at the time of surgery.
Methods: The culture results from 193 consecutive shoulder arthroplasty revisions for pain or stiffness, but without clinical evidence of infection, were correlated with a host of preoperative and intraoperative observations. Univariate and multivariate logistic regression models were used to identify factors predictive of a positive culture for P. acnes or other organisms.
Results: One hundred and eight of the 193 surgeries had positive cultures; 70% of the positive cultures grew P. acnes. While 97% of the P. acnes cultures were positive by 3 weeks after surgery, only 45% were positive after one week. The prognosis for a positive P. acnes culture was statistically significantly increased for male patients, shoulders with humeral loosening and osteolysis on preoperative x-ray, surgical findings of glenoid wear, osteolysis membrane formation and cloudy fluid,
as well as cases in which there was a surgical suspicion of infection. We developed a multivariate model to predict P. acnes with 92% sensitivity and 47% specificity.
Conclusions: Preoperative and intraoperative factors can be used to prognosticate the risk of a positive culture for P. acnes. This evidence may be useful in decision making at the time of revision shoulder arthroplasty before the definitive culture results become available.
Thus, the diagnosis of infection is usually made from culture at the time of revision surgery where multiple samples are taken along with frozen sections. This is why it is so important to avoid administering antibiotics until multiple cultures are obtained to minimize the risk of sampling errors. For this reason we do not start antibiotics after an aspiration in the office or emergency room because of the possibility that this culture may not yield a representative result (i.e. it may be falsely negative or contaminated by skin flora).
If the infection is acute, the organism sensitive to antibiotics, and the patient healthy, we may elect a vigorous debridement of soft tissue inflammation, a surgical scrub of the joint surfaces, and irrigation with copious volumes of antibiotic saline solution. If the infection is established, we will usually remove all components and cement and then replace only an uncemented humeral component, smoothing the residual glenoid surface if needed. We no longer use antibiotic impregnated spacers because (1) they seem no more effective in resolving infection than a primary exchange and (2) they obligate the patient to a revision procedure which is usually not necessary in a primary exchange.
Culture-specific intravenous antibiotics are used for a minimum of six weeks.
Revision surgery due to stiffness – unwanted bone and overstuffing
The diagnosis of stiffness is made by the finding of reduced humeroscapular rotational laxity in flexion, cross body adduction, internal and external rotation with the arm at the side, and internal and external rotation with the arm in 90 degrees of abduction. Some cases of stiff shoulder after arthroplasty can be managed with soft tissue releases alone. Often, however, soft tissue releases may be insufficient because of technical problems with the implants. High quality anteroposterior and axillary radiographs are needed to determine the presence of unwanted bone [See Figure 24], displaced tuberosities [See Figure 25] or other ‘hard’ causes of restriction of motion. As shown previously, all adhesions in the humeroscapular motion interface are lysed. A 360-degree release of the subscapularis is performed freeing it from the coracoid the coracoid muscles the axillary nerve and the glenoid lip. The capsule is released around the periphery of the glenoid – 360 degrees unless there is posterior instability in which case the release is stopped at 190 degrees.
All unwanted bone such as residual osteophytes between the medial humerus and inferior glenoid is removed. If the greater tuberosity is malunited posteriorly, it can block external rotation. We prefer to avoid tuberosity osteotomy unless it is absolutely necessary because of the difficulties of mobilizing the tuberosity and of obtaining a secure tuberosity union to the shaft after a humeral arthroplasty. For this reason, we prefer to resect prominent tuberosity, bone leaving the cuff intact if this is at all possible.
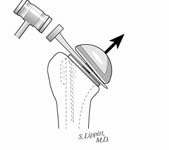
If the joint is overstuffed (the components consume too much of the joint volume) as shown below [See Figure 26], the intraarticular prosthetic volume may be reduced by removing or revising the glenoid component and by reducing the height (thickness) of the humeral component.
Varus positioning of the stem can also result in overstuffing and require component revision. [See Figure 27].
More often than not a combination of soft tissue, bony, and component procedures are required to optimize the motion in a stiff arthroplasty.
Click an image below to enlarge and view in the gallery.
Revision surgery due to humeral fracture
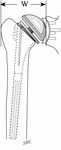
Fracture of the humeral shaft is a recognized cause of shoulder arthroplasty failure. The risk for such a fracture is increased in individuals with poor bone quality or with an increased risk of falling because of poor eyesight, balance problems, confusion, or muscle weakness. The risk is also increased when there has been weakening of the humeral shaft from reaming as explained in a previous post or from loosening of the humeral component. Our experience indicates that humeral fracture risk is lessened when endosteal reaming is minimized and when impaction autografting is used to secure the stem in the humeral shaft.
The diagnosis of shaft fracture is revealed by high quality anteroposterior and lateral views that span the entire humerus.
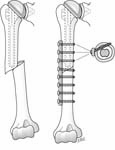
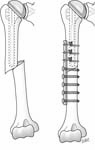
If the component is not loose, it can be difficult to remove in the presence of a fracture. In this situation we prefer to leave the prosthesis in place and fix the fracture using a plate placed so that the screws in the proximal fragment pass through the cortical bone either anterior or posterior to the stem of the prosthesis. [See Figure 28]
If good fixation in the proximal fragment cannot be achieved with screws, circlage around the plate may be used, but extreme care must be used to avoid circlaging the radial nerve posteriorly and to avoid crushing the bone with excessive tightening of the wires. [See Figure 29]
If the humeral prosthesis is loose it may be removed and replaced with a long stem prosthesis fixed with cement or bone graft and supplemented with circlage fixation again taking care to protect the radial nerve. [See Figure 30]
In all cases of humeral fracture we prefer to use autogenous bone graft or cancellous allograft around the fracture site.
Click an image below to enlarge and view in the gallery.
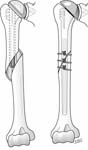
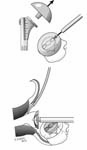
Revision surgery due to glenoid component failure – wear or loosening
The glenoid component of a total shoulder may fail because of wear (as shown in Figure 31) or because of loosening with radiolucent zones seen in black on x-ray (as shown in Figure 32).
If the glenoid is worn but securely fixed, it is removed by first cutting the pegs or keel and removing the glenoid component face, allowing access to the bone surrounding the fixation, so that the remainder of the component and the cement can be dissected from the glenoid bone. [See Figure 33]
If the glenoid component is loose, it can be easily pried out of the glenoid bone. [See Figure 34]
We will discuss the management of the glenoid after component removal in the next section.
Click an image below to enlarge and view in the gallery.
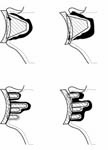
Revision Surgery due to glenoid component failure – loss of supporting bone
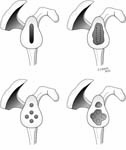
To continue with our discussion of the management of glenoid component failure we’ve observed that attempts to reinsert a new glenoid component are accompanied by a high failure rate due to to the loss of supporting bone (two right hand figures from See Figure 35) in contrast to the situation when the first glenoid component was inserted (two left hand figures from See Figure 35).
Thus, when we encounter a substantial glenoid defect (such as that shown in See Figure 35) rather than using a large amount of cement bone graft or a special component to fill the defect, we have been pleased with the result from removing all polyethylene bone cement and rough bone and then contouring the residual glenoid bone to support a new humeral head component (usually one with a diameter of 56 mm to achieve the maximal contact area). This essentially converts the failed total shoulder to a ream and run. We do not fill the defect with bone graft finding that the defect fills in itself with time.
Click an image below to enlarge and view in the gallery.
Revision surgery due to glenoid component failure – instability
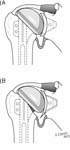
What if a failed shoulder arthroplasty is associated with instability that is unwanted translation of the humeral head on the glenoid? Such a shoulder requires a careful history examination and radiographs. The physical examination of strength in isometric internal rotation with the arm against the abdomen, isometric elevation of the internally rotated arm, and isometric external rotation of the neutrally rotated arm at the side, as well as expert shoulder ultrasound, can evaluate the integrity of the subscapularis, supraspinatus, and infraspinatus respectively. High quality and appropriately oriented anteroposterior and axillary radiographs will reveal the glenohumeral relationship (including the superior/inferior and anteroposterior relationship of the center of the humeral head and the center of the glenoid), the integrity of the tuberosities, the orientation of the glenoid, and much about the type and position of the humeral component. If knowledge of the humeral version is essential, it may be necessary to perform an examination under fluoroscopy, noting the rotational position of the arm that places the humeral neck in greatest profile.
In terms of treatment options here are some to consider.
Subscapularis deficiency may not be reconstructable. If there is good quality muscle and tendon, a repair may be possible after a complete release of the medial muscle and tendon. A hamstring autograft may be useful for extending the tendon length.
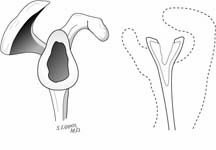
If the glenoid component is in excessive anteversion, its intrinsic balance stability angle does not provide anterior stability due to its misalignment with the net humeral joint reaction force (determined principally by the scapular origin of the scapulohumeral muscles). This etiology of anterior instability is suspected when there is minimal resistance to the anterior load and shift test. The diagnosis of glenoid component anteversion can be made by examination of a true axillary view of the joint. The glenoid centerline normally projects out the anterior scapular neck at the centering point. In the anteverted glenoid, the glenoid centerline projects down the scapular body or behind it. When anterior instability is associated with glenoid anteversion, reorientation of the prosthetic glenoid centerline is usually indicated. Because the anterior glenoid lip of a polyethylene component is usually worn by the recurrent instability, the prosthesis often needs to be changed. Thus, the revision is accomplished either by removal of the prosthetic component and performance of a properly oriented, non-prosthetic glenoid arthroplasty or by reinsertion of a glenoid component with its centerline in proper orientation with the scapula. (See Figure 37)
If the anterior glenoid bone stock is deficient, an iliac crest bone graft may be secured to the anterior glenoid and then reamed either for a non-prosthetic glenoid arthroplasty or to fit the back of the glenoid prosthesis. (See Figure 38)
If the glenoid component is in excessive retroversion, its intrinsic balance stability angle does not provide posterior stability. (See Figure 39)
Clinically, the shoulder will demonstrate diminished resistance to posterior load and shift and instability on cross body adduction. On the axillary radiograph the glenoid centerline is seen to project through the bony glenoid more anteriorly than the normal centering point. As for glenoid anteversion, correction may include re-establishing the normal glenoid centerline and then performing corrective reaming for a non-prosthetic or prosthetic glenoid arthroplasty. (See Figure 40)
At times it may be necessary to use a posterior bone graft, but it is challenging to obtain secure fixation of the graft to the host glenoid bone. (See Figure 41)
If there is insufficient glenoid bone for a reconstruction and if an iliac crest autograft cannot be performed because of insufficient quality bone stock to which it can be anchored, a glenoidectomy can be considered as a salvage procedure. In this procedure the residual glenoid is resected down to the level of the scapular spine. An appropriately sized humeral head prosthesis is inserted to articulate with the glenoid neck, the scapular spine, and the base of the coracoid. (See Figure 42)
Click an image below to enlarge and view in the gallery.
Revision surgery due to superior instability
Loss of the integrity of the coracoacromial arch can be a major problem following shoulder arthroplasty, allowing anterosuperior escape of the proximal humerus from the glenoid. This problem is often caused by prior surgery attempting to repair a large rotator cuff tear. It is most commonly seen when an acromioplasty has been performed, compromising the stabilizing effect of the coracoacromial arch. (See Figure 43)
In this situation a reverse total shoulder arthroplasty may be considered.
Revision surgery for humeral component revision
Removal of a Humeral Implant
In some instances removal of the head of a modular component is sufficient to accomplish the desired revision including gaining access to the glenoid. (See Figures 44-45)
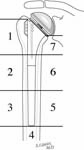
Often, however, revision requires removal of the entire humeral component. Extraction of a humeral prosthesis can be straightforward in the case where there is radiolucency in all 7 zones between the humeral endosteum on one hand and the prosthesis and any cement used to fix it on the other. (See Figure 46-47)
On the other hand, removal of a humeral prosthesis can be extremely challenging, for example, when it has been thoroughly cemented to the bone of an osteopenic humerus or when an ingrowth or textured surface humeral component has been used. Before embarking on the removal of a cemented humeral component, the difficulty and the necessity of the removal of cement needs to be anticipated. The need for cement removal is influenced by the presence or absence of infection, the requirement to change prosthesis size and position, and the extent of the cementation. In the absence of infection and when the cement is secure to the bone, we will often opt to work within the previous cement mantle (for example using a component with a smaller diameter stem and recementing within the old cement) rather than running the risk of removing it. The anticipated difficulty and the possibility of fracturing the humeral shaft or tuberosities during the removal are discussed with the patient in detail preoperatively. The surgical inventory is carefully reviewed to assure that long stem implants of the appropriate diameters and head sizes are available for the subsequent reconstruction.
Our approach to prosthesis removal begins with the removal of soft tissue bone ingrowth and cement from around the humeral head or, in the case of a modular prosthesis, from around the collar and from around the fins of the prosthesis. (See Figure 48)
If the prosthesis cannot be removed easily at this point, enough bone in the area of the bicipital groove is cut to allow the positioning of a bone tamp parallel to the shaft with one end beneath the collar or head (See Figure 49)
The elbow is flexed to 90 degrees and the arm is stabilized to the thorax while the surgeon strikes the bone tamp so that a longitudinal impact is applied to the proximal prosthesis along the axis of the humeral shaft. If reasonable impact does not dislodge the prosthesis, a longitudinal humeral osteotomy is started in the bicipital groove.
When the bone is cut, the osteotome is twisted slightly to open up the endosteal cross section of the humerus. Impact is applied as before with the bone tamp. The linear osteotomy is continued sequentially with the osteotome twist each time until the prosthesis can be removed. (See Figure 50)
If cement removal is necessary this can be performed with the usual cement removal tools inserted down from the canal opening at the proximal humerus or through the humeral osteotomy. In our hands it seems more safe and effective to monitor the integrity of the bone by extending the incision sufficiently inferiorly so that the bone can be palpated during the cement removal rather than relying on intra-operative fluoroscopy. In any case, burrs and osteotomes tend to cut the often thin and soft bone preferentially to the hard cement; thus the surgeon must be prepared for bone penetration and its possible consequences (nerve damage, additional fracture, leakage of cement).
If a secure cement mantle remains after humeral component removal, the new humeral component with a smaller diameter can be cemented into the old mantle.
If a cementless reconstruction is desired, the humerus can be reassembled using a long stem prosthesis press fit as far down the distal humerus as possible. (See Figure 51)
At this point in the case the medullary canal can be divided into two components – the proximal section that was opened to retrieve the prosthesis and the distal aspect consisting of an intact cylinder. The fixation of the prosthesis depends on this distal segment, especially in the circumstances where a cementless reconstruction is desired, for example, after the debridement of an infected arthroplasty or because of concern regarding an adverse reaction to methylmethacrylate. In this situation the cylindrical distal humeral segment is reamed with cylindrical reamers until the fit and fill of a cylindrical component stem is optimized. Since the quality of the fit depends on the length of the bone-prosthesis contact, the length of the prosthesis inserted into the cylindrical segment is maximized. This is accomplished by extending the reaming as distally as possible and by maximizing the length of the prosthetic stem. We often start with a 300 mm stem of the proper diameter insert it fully in the reamed canal and then measure how much of it needs to be trimmed for the articular surface to be in proper register with the glenoid. The stem is then cut with a high-speed motorized disk and smoothed of any burrs remaining from the cut. The prosthesis is then impacted into the distal cylindrical segment paying particular attention to the version. The open proximal humeral segment is then folded around the prosthesis. Because the posterior and medial periosteum and muscle attachments have been preserved, the osteotomized bone can be reconstructed by suturing the osteotomy closed using drill holes on either side (the ‘bodice’ repair). (See Figure 52)
Circlage can also be used but care must be taken to protect the radial nerve that can be circlaged in its musculoskeletal groove.